Glass Manufacturing: Part One

Glass is produced for the packaging industry as a container for perfume, alcoholic and non-alcoholic beverages, jarred foods, pharmaceuticals, and more. Most of us have a really basic understanding of how glass is made: Sand and other raw materials are melted at high heat, formed in molds, and when it cools—BINGO! Glass! In this lesson we’ll explain that process in a little more depth.
Learning Objectives
By the end of this lesson, you will be able to:
Explain how raw materials are prepared and mixed to form the glass batch.
Describe how raw materials are melted and refined into molten glass.
Glass Making Overview
All commercial glass is made from a silica base sourced from high-purity sand. Limestone, soda ash, and alumina are added to make soda-lime glass, which is the most common type of glass for commercial bottles and jars. Cullet, or broken glass collected from plant operations and post-consumer waste, also makes up a percentage of the new glass composition. This is added to enhance the melting rate and reduce overall plant energy requirement.
The manufacturing of glass has three primary parts:
The Batch House
The Furnace (which will be covered in this lesson)
The Forming Process (which will be covered in the next lesson)
The Batch House
The batch house is the initial location where raw glass material is inspected, weighed, and stored before entering the glass furnace.
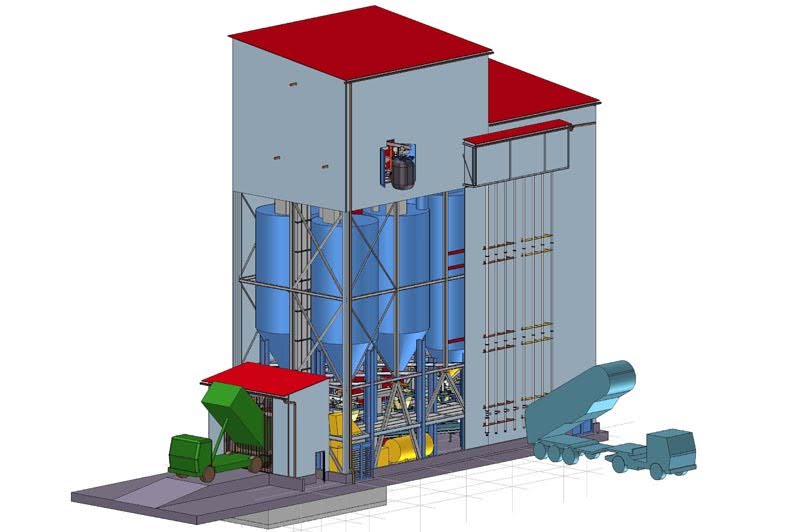
Raw materials arrive at the batch house by the truckload and are inspected and sorted (both mechanically and visually) before being stored in silos.
Materials are carefully weighed out according to the required “recipe” and then sent to the mixer, located just above the furnace.
A typical batch contains about a ton of sand and the appropriate ratio of soda ash, limestone, and other mineral compounds and conditioning materials.
Coloring agents can be added before entering the furnace or at the forehearth.
In some plants, cullet is added after the raw materials are mixed to minimize wear and tear on the mixer.
Water is often added to the mixer just before the batch is sent to the furnace via belt conveyor or monorail.
This decreases the possibility of the batch putting off dust or de-mixing during transport, and increases the efficiency of batch melting.
The Furnace
Next is the furnace. Most commercial glass plants operate 24/7, and a well maintained furnace can last for as many as 10 years. Over that lifetime, large furnaces will produce up to 1,000 tons of glass for every meter of internal surface area.
Furnaces will be rebuilt every 10–15 years at a cost of $8–$10 million.
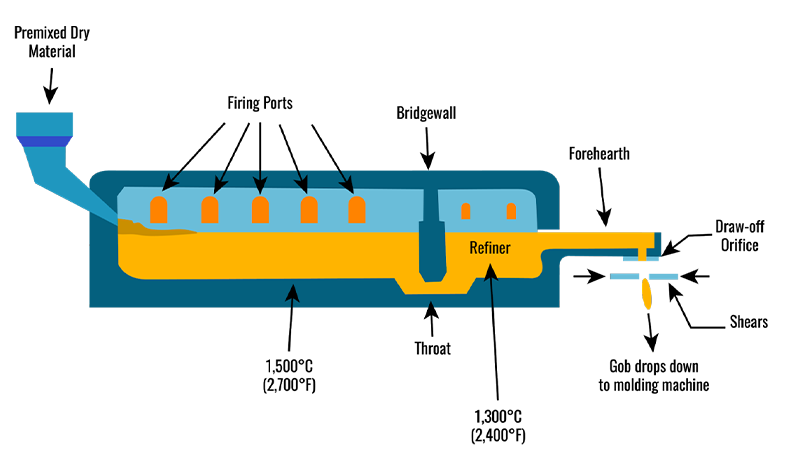
Commercial glass is made in a gas-fired melting or fusion furnace, lined with high-temperature refractory materials.
The premixed raw materials are continuously fed into one end of the furnace, which, at 1,500ºC, is known as the “hot end.”
The batch progresses to the melter, where the actual melting takes place.
Inside the melter, 3–7 ports direct combustion and exhaust gases over the batch.
This process initiates the release of carbon dioxide gas from the decomposition of carbonate ingredients.
The released gases and the convection currents in the furnace help to mix the glass.
The raw materials fuse into glass at about 1,510°C or 2,750°F.
Most furnaces are designed to use natural gas, but oil, propane and electricity can also be used to power them.
In air-fueled furnaces, the high temperatures create environmentally-objectionable nitrogen oxides as an off-gas.
Newer furnaces use oxygen as a cleaner fuel instead of air to eliminate a possible pollution source and to reduce the overall energy requirement up to ⅓.
The hot flue gases pass through a heat exchanger and lower energy costs for the manufacturer by heating incoming cold air.
Smaller furnaces are used to produce special glassware for applications like fancy cosmetic bottles, special colors, or decorative glassware.
The melted glass passes from the melter to the refiner through the throat, a water-cooled tunnel that extends through the bridge wall.
The refiner is a holding basin where the glass batch cools to a uniform temperature before entering the forehearth.

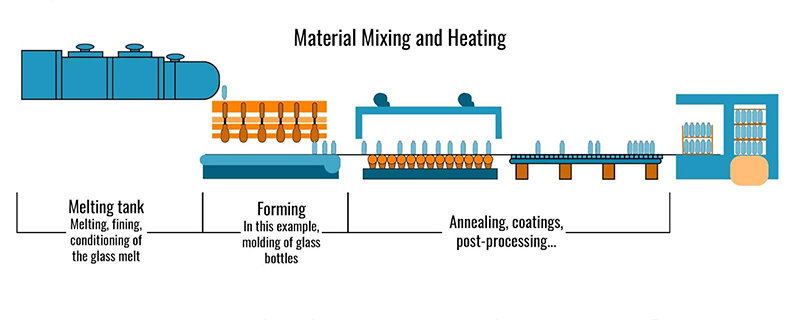
Material Mixing and Heating
The process of material mixing and heating has been visualized in this graphic.
In the next lesson, we will continue this overview of glass manufacturing by diving into the forming process, mold types, annealing, and more!
By signing up you indicate you have read and agree to our Terms of Use. Packaging School will always respect your privacy.